CAREER HIGHLIGHTS:
2013 - 2017 IRISS Medical Technologies Ltd.
VP Engineering

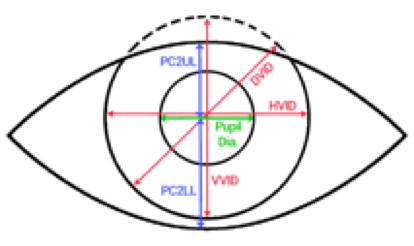
Eye Check, IRISS’s first major product, was based on the automated red eye correction software created during my time at Pixology. The core red eye detection technology was extended so it could be used to automatically measure eye features such as pupil, iris, aperture and deviation. This was pretty radical because, to date, optometrists, etc measured these things manually using a millimeter ruler. The best practitioners are able achieve an accuracy of 0.5mm using a ruler. The IRISS device can give any user measurements accurate to 0.1mm.

After discovering that there was just one Android smartphone that fitted the technical needs of the software, we went on to find that it was only available in the Far East. I spent lots of time in Taiwan, working with the manufacturer to make the handset suitable for use as a medical device in the EU. In addition, we designed a housing that incorporated a “fixation target” (flashing lights for the patient to look at) in order to make the entire device more user friendly and easy to handle. It wasn’t as straightforward as it sounds...


The second
prototype
looked as
Heath-
Robinson
as the first.
But at least
the button
didn’t fall off
every five minutes.
Having said that, the
first prototype was
the one that all the real
users tested the software with.
The third prototype saw a proper housing
for a purpose-made fixation
target.

This was the first Eye Check
hardware prototype. For the fixation target, a battery-driven flashing LED star was controlled by a push-switch stuck to the side with Blu-Tack. A lot of the testers really liked it!
But
because the button
was on the same side as
the shutter, nobody could cope with using the
target and taking a picture at the same time.

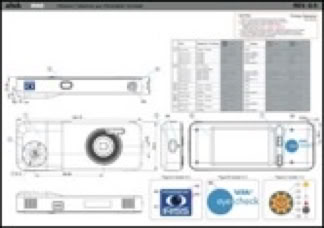

After a few iterations of the housing that enclosed the smartphone in varying degrees, hours and hours of software testing & bug fixing,
the help of an experienced team of industrial design engineers and a precision manufacturing facility, the first release of the Eye Check device was born.
During one of my visits to Taipei, one of their technical guys and I came up with a very novel way to figure out what distance the patient was from the device when the picture was taken.
We did this by modifying the camera-module firmware so it added the focus stepper-motor position to the image metadata. We then created a calibration process that generated a look-up table for each device and a tiny firmware service which added the actual distance in mm to the metadata as well. The Software application then used that distance to calculate the other dimensions in the picture. Problem solved!


Regulation is a very important aspect of medical device design, development, manufacturing, distribution, sales, support and reporting. In order to sell our devices in the US and Europe we needed to comply with EEC, FDA and FCC regulations. A prerequisite to claiming that compliance was for the company to be ISO13485 certified. Achieving all of this, from scratch, is a huge task. I was the lead in getting full EU & US certification in place in less than 12 months from starting the process.

My name is Iris
You’ll find me around
whenever testing is being
done or there are devices to be calibrated. I am the perfect test subject because I’m available 24/7, my appearance never changes and I never, ever get bored. I also have a pretty good squint so I can even help
to test deviation.

We made an iPhone app too.


Copyright © 2010 - 2026 Mike Stroud